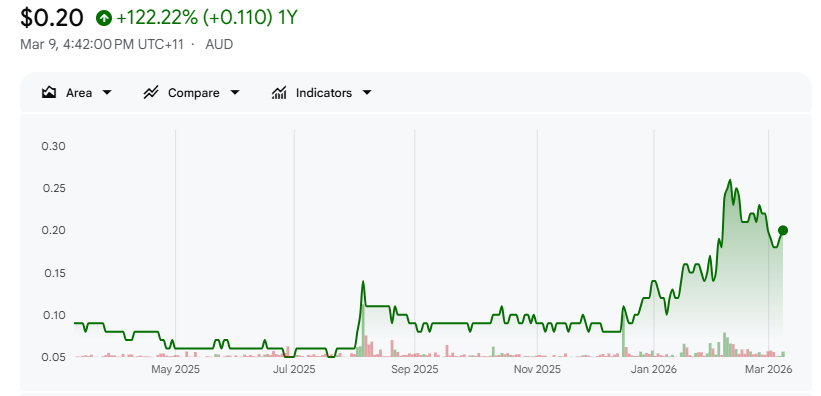
Rhythm Biosciences (ASX: RHY)Share Price and News
Overview of Rhythm Biosciences
Rhythm Biosciences (ASX: RHY) is a Melbourne-based cancer diagnostics company focused on improving patient outcomes through earlier and more accurate cancer detection. The company operates two complementary products: ColoSTAT, a blood-based test for the detection of colorectal cancer, and GeneType, an AI-driven polygenic risk scoring platform that helps individuals understand and manage their personal risk across multiple cancers and chronic diseases.
ColoSTAT uses antibodies to measure five specific protein biomarkers in a patient's blood sample, generating a colorectal cancer risk score that a GP can use to guide clinical decision-making. The most recent clinical study demonstrated 81% sensitivity, 91% specificity, and a 99% Negative Predictive Value, meaning that a negative result carries a 99% probability that the patient does not have colorectal cancer.
GeneType uses a simple cheek swab to assess polygenic risk across multiple factors including genetic variants, BMI, lipid levels, breast density, blood pressure and cholesterol, generating a personalised risk profile across six cancer types: breast, prostate, colorectal, ovarian, melanoma, and pancreatic. GeneType is expected to expand to additional indications over time, including potential application in Type 2 diabetes risk prediction.
Together, those two products address a combined global addressable market that Rhythm Biosciences estimates at approximately 500 million individuals worldwide.
Rhythm Biosciences' Company History
Rhythm Biosciences listed on the ASX in 2017, having acquired the ColoSTAT intellectual property which originated from research conducted at the CSIRO from approximately 2003 onwards. The CSIRO's foundational work identified 68 proteins that vary in concentration between patients with and without colorectal cancer, then narrowed those down to the ten most likely to detect the disease.
Rhythm Biosciences built on this foundation to develop a viable test kit, and subsequent validation studies (Studies 6 and 7) further simplified the biomarker panel to five proteins and demonstrated strong clinical performance.
In 2023, the company submitted a TGA registration application for ColoSTAT via the conventional pathway. The application was withdrawn following regulatory feedback, not due to any issue with the test's clinical efficacy, but because the company did not yet have sufficient independent production batches to satisfy the regulator's requirements at that time. This setback delayed commercialisation but did not undermine the technology itself.
In late 2024, Rhythm Biosciences made a transformative acquisition when the previous owner of GeneType, Genetic Technologies Limited, entered administration. Rhythm acquired the GeneType business for just over A$600,000, with the deal including the intellectual property, existing customer contracts, patient data, personnel and commercial infrastructure. The acquisition was completed ahead of schedule. First commercial sales from GeneType were recorded in early 2025, carried over from legacy arrangements under the previous owner's commercial relationships.
Since completing the acquisition, Rhythm Biosciences has moved on multiple fronts simultaneously. In late 2025, the company established ISO 15189:2022 accreditation for its Melbourne laboratory and began offering ColoSTAT as a clinical laboratory service to selected clinical leaders from December 2025. This accredited laboratory pathway mirrors the CLIA pathway that Lumos Diagnostics (ASX: LDX) has pursued for commercialisation in the United States.
In parallel, the company progressed several key GeneType distribution partnerships: a strategic pilot with a leading Australian pathology provider, a collaboration with Memorial Sloan Kettering Cancer Centre supporting studies in breast and prostate cancer risk, integration with the Know Your Lemons mobile application (which has a global reach of more than 2 billion people online and operates education programs in over 75 countries), and a partnership with Catch Bio, a cancer prevention platform that enables individuals to identify and manage their cancer risk.
Rhythm Biosciences' Future Outlook
Rhythm Biosciences enters 2026 with greater commercial momentum than at any point in its history, and Pitt Street Research considers this the year in which both ColoSTAT and GeneType begin to demonstrate meaningful commercial traction.
The immediate priority for ColoSTAT is completing the NATA variation to scope assessment, which would formally add ColoSTAT to the company's accredited laboratory services rather than offering it as a general laboratory service. This step would support broader clinician use and a more formal commercial rollout beyond the initial limited programs that commenced in December 2025. Looking further ahead, the company plans to resubmit to the TGA for formal ARTG inclusion as an In Vitro Diagnostic Medical Device, which would expand access to non-laboratory environments. The company is also in the early stages of international expansion: the NHS Southern Hub Research Team in the United Kingdom will evaluate ColoSTAT for suitability for general use during 2026, and the company is developing a lung cancer screening assay with pre-clinical data evaluation and assay prototype design expected to be completed during CY26.
For GeneType, the company has committed to significant quarter-on-quarter revenue growth, including over 100 percent growth in Q1 of FY26. The sales pipeline is growing rapidly, underpinned by the four distribution and collaboration partnerships established in 2025. The GeneType test is currently priced at A$399 per risk test kit and is already generating revenue, establishing a commercial foundation that ColoSTAT has yet to match.
The market opportunity backing both products is substantial. The Australian screening population for colorectal cancer at 7.4 million people aged 50 to 74, with 96.7 million in the same bracket in the United States and 20.8 million in the United Kingdom.
For GeneType, the company targets a global market of 207 million Normal Active Risk Assessors and 69 million Higher Risk assessors. Even a 1 percent penetration of that combined addressable market would represent approximately 2.76 million patients.
The underlying disease burden reinforces the opportunity. Colorectal cancer was estimated to have caused 1.9 million new cases and 900,000 deaths globally in 2022. These figures are projected to grow to 3.2 million cases and 1.6 million deaths annually by 2040 if current trends continue.
Participation rates in Australia's government-funded Faecal Immunochemical Test program remain at only 40 to 45 percent, largely due to the cumbersome and unpleasant nature of stool collection. Research has shown that blood tests are the preferred screening method for more than 83 percent of currently unscreened individuals, and at least one study found that uptake increased from 12.6 percent to 93.5 percent when a same-day blood test replaced the FIT as the offered screening option.
This represents the core commercial case for ColoSTAT: a clinically validated blood test that addresses the primary reason why millions of people avoid bowel cancer screening.
Is Rhythm Biosciences (ASX: RHY) a Good Stock to Buy?
We believe the investment case rests on several genuinely differentiated factors. ColoSTAT has over a decade of validation work behind it, originating from foundational CSIRO research, and its clinical performance compares favourably to the government-funded FIT in both sensitivity and patient convenience. GeneType is already generating revenue and was acquired for a price that the market has largely not yet recognised. Case in point: the company's market capitalisation barely moved following completion of the GeneType acquisition, suggesting the market continues to value the company as if GeneType does not exist. For investors who look closely at both assets, we believe there is a meaningful valuation gap.
The risks, however, are real and must be clearly understood. The company is not yet profitable and will likely require further capital raisings to guide both products to self-sustaining commercialisation. Regulatory risk remains present: ColoSTAT's TGA pathway has already been disrupted once, and any further setbacks in achieving NATA accreditation or a future ARTG inclusion would materially delay the commercial timeline. Commercial execution risk is significant given that both products are relatively early in their market development, and the company must successfully convert its growing partnership network into recurring revenue. Key personnel risk is also acknowledged given the degree to which the company's strategy relies on the current leadership team.
For investors seeking exposure to cancer diagnostics with a credible near-term commercial pathway, Rhythm Biosciences presents a genuinely compelling opportunity at current valuations. CY26 is expected to be the year that commercial revenues from both products begin to close the gap between market price and fundamental value, and the pace of that revenue ramp will be the key metric for investors to monitor.
Our Stock Analysis
Breville (ASX:BRG): Will consumer demand for $4500 coffee machines offset the impact of Trump’s tariffs?
Breville (ASX:BRG) is one of those consumer stocks caught in the crosshairs of Trump’s tariffs and fell 30% in 2…
What are the best ASX Consumer Stocks for FY25? Here are our top 5 picks!
It is time to reveal our best ASX Consumer Stocks for FY25! We will be addressing both discretionary and staple…
Our 5 best ASX stocks for 2024
It is nearly the end of 2023, and so it is time to share our best ASX stocks for 2024!…
The 4 Best ASX stocks for October!
In this article, we look at the best ASX stocks for October. This month we have four choices and underpinning…
Our 5 favourite ASX consumer discretionary stocks!
ASX consumer discretionary stocks have fared terribly over the last 12 months. 4-decade high inflation and rapidly rising interest rates have…
Frequently Asked Questions
Rhythm Biosciences (ASX: RHY) is a Melbourne-based cancer diagnostics company that develops and commercialises blood-based and genomic tests for earlier cancer detection and risk assessment. Its two products are ColoSTAT, a blood test for colorectal cancer detection, and GeneType, an AI-driven polygenic risk scoring platform that assesses personal risk across six cancer types using a simple cheek swab. Together, the two products address a combined estimated global market of approximately 500 million individuals.