Prescient Therapeutics (ASX:PTX): It has taken the plunge into a Phase 2 trial against T-Cell Lymphoma

There’s plenty of ASX oncology biotechs conducting clinical trials, but Prescient Therapeutics (ASX:PTX) is one of the closest to commercialisation. The company has just started Phase 2 for T-Cell Lymphoma and successful data here could suffice to appease the FDA without a Phase 3 trial, given the lack of treatment options currently available, but also because of how efficacious it has been shown to be. Success at the current stage would be good news not just for investors, but to sufferers of this terrible cancer.
What are the Best ASX Stocks to invest in right now?
Check our ASX stock buy/sell tips

Prescient Therapeutics’ journey
Prescient Therapeutics originated with PTX-100 and PTX-200 in 2014. Prescient listed on the ASX in a reverse shell transaction using the shell of a failed drug developer called Virax. PTX-200 is on the backburner, but PTX-100 is in a pivotal clinical trial. PTX-100 is small molecule that can attack cancer by disrupting a cellular signalling pathway called ‘Ras’.
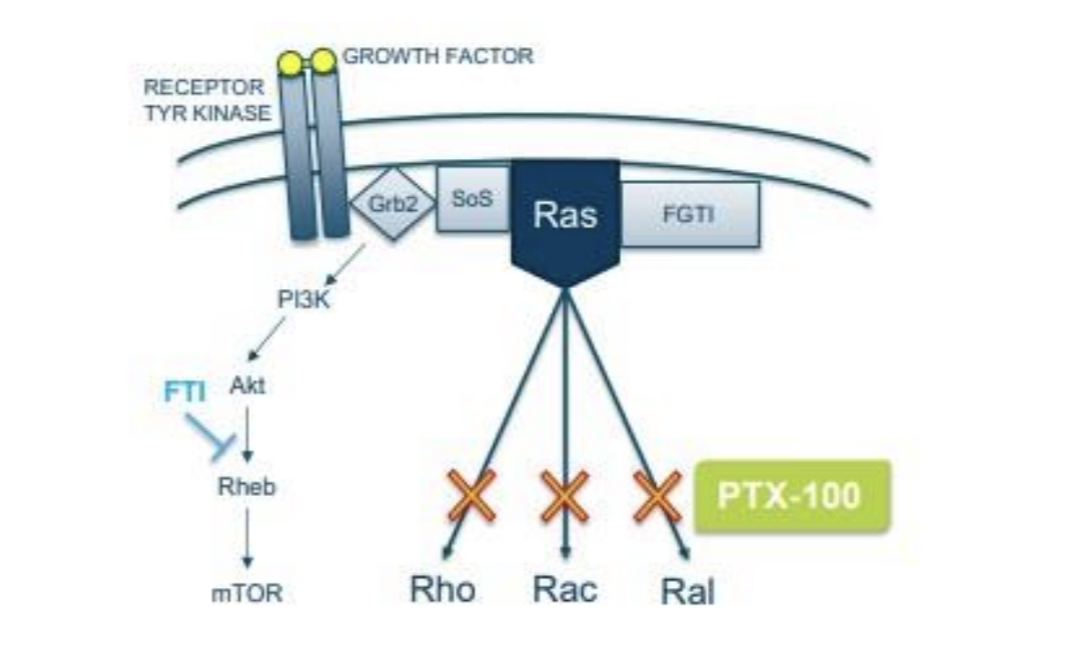
Source: Company
Over the last half-decade Prescient has gathered some great data on PTX-100 in T-Cell Lymphomas. T-Cell Lymphoma is a rare but aggressive cancer with a low survival rate. While only around 5,000-6,000 people a year will be diagnosed with a T-Cell Lymphoma in the US the market opportunity could be in the billions, if Acrotech Biopharma’s Folotyn with its $842,585 per patient per year price tag is any guide.
In 2019 Prescient started a Phase 1 study of PTX-100, initially as a dose escalation study in a variety of tumour types. Phase 1b completed in July 2021 with an excellent safety profile and a clinical signal in T-Cell Lymphoma. An expansion cohort in T-Cell Lymphoma opened in April 2022 to focus on T-Cell Lymphomas and by October 2022 Prescient was reporting favourable clinical activity in this cohort. In July 2022 PTX-100 was granted Orphan Drug Designation by the US FDA for Peripheral T-Cell Lymphoma and for all T-Cell Lymphomas in March 2023.
By March 2023, 40% of patients in the trial responded and seven of 10 had durations of response exceeding the standard of care. And it is important to keep in mind that this specific cohort represented ‘relapsed and refractory patients’ – in other words, patients for whom previous treatments had either not worked at all or only temporarily. By December 2023, there was a Clinical Benefit Rate of 66% – patients who either saw complete eradication of the disease or some clinical response.
Now what?
The next step for PTX-100 was a Phase 2 trial, and the company started this in May 2025. The study will focus on Relapsed and Refractory Cutaneous T-cell lymphomas, meaning it will be faster and smaller than had it been a broader TCL trial. It will occur across 15 sites globally and occur in 2 parts. The first will enrol 40 patients and determine an optimum dose for the next stage.
Then there’ll be an efficacy and safety determination in 75 patients. Eligible patients will need to have received and failed at least two prior lines of systemic therapy. The primary endpoint will be the objective response rate (in other words, patients with complete or partial responses).
PTX hopes that this trial will be a success and that if so, PTX-100 could be submitted for FDA approval if and when it passed this study without the need for a Phase 3 study. Assuming success in all regards (FDA approval to the Phase 2 trial to be pivotal, successful Phase 2 results and FDA approval for commercialisation), commercialisation could occur in as little as 2-3 years from now. PTX aims to initially seek approval for CTCL and then leverage this for a separate PTCL (Peripheral T-cell lymphoma) registration study.
As of late October 2025, nearly half of the planned sites have been initiated (7 of the planned 16). Six patients are in dosing with further patients in the screening process. One of the US sites is one of America’s top cancer research centres, the VCU Massey Comprehensive Cancer Centre. PTX closed the September quarter of CY25 well capitalised for the medium term with $12.3m in cash (up from $6.9m 3 months prior) thanks to a successful capital raising.
Other potential catalysts exist
While the trial will be the most important determinant for the creation of shareholder value, it will not be the only one. Nonetheless, it is not unreasonable to imagine there could be M&A or partnering interest for this drug – the latter could occur even prior to commercialisation as can happen with orphan drugs.
Given that developers of orphan drugs are able to obtain substantial benefits (such as tax credits, long-term periods of market exclusivity and even waiving of certain fees) and can also fetch premium prices (several hundreds of thousands of dollars for example), big pharma can pay a premium.
One example Horizon Therapeutics – it has Tepezza, the only approved treatment for thyroid eye disease, and was bought by Amgen’s in October 2023 for a staggering US$27.8bn. This followed over $3.5bn in sales in the first two full years on the market (2021 and 2022). Yes, a figure of US$27.8bn might be too high for PTX at this stage, but companies could pay a valuation 1.5-3x PTX’s current market capitalisation and still get a potential bargain.
We may also see partnering or licensing deals prior to commercialisation as we saw with Dimerix (ASX:DXB). Dimerix demonstrates that it is not impossible for deals to be struck prior to approval and even during the pivotal clinical trial. Such a deal would not only facilitate royalties on sales, but likely provide for payment of certain R&D and commercialisation costs as well as potential upfront payments that could help fund development of OmniCar and CellPryme. Alternatively, we could see partnership opportunities in the sense of using PTX-100 as part of drug combinations.
CellPryme and OmniCar have potential too
PTX has also been progressing its work with CellPryme and OmniCAR, both platforms to assist with CAR-T (Chimeric Antigen Receptor T-Cell) therapy. CAR-T therapy is a therapy that utilises the body’s own T-cells, which are isolated from the patient’s peripheral blood, endowed with enhanced specificity and killing efficacy towards the patient’s cancer cells, and then reinjected into the host, where they aid in tumour clearance.
The treatments on the market today show very encouraging remission rates of over 80%. Many CAR-T therapies, however, remain inaccessible to many due to high costs and have some challenges holding them back, particularly efficacy, durability of CAR-T cells, multi-targeting and post-infusion control. This is where CellPryme and OmniCAR come in.
CellPryme is a cell therapy enhancement platform, with two applications (CellPryme-M and CellPryme-A) that can be used separately or concurrently with significant power together and in their own right. CellPryme-M, through the introduction of molecules during a cell manufacturing process, allows better kinds of cells to be produced, which improves cellular therapies like CAR-T. CellPryme-A is an intravenous drug administered alongside cellular immunotherapy and allows the tumour microenvironment to be overcome.
OmniCAR, in simple terms, operates as an immune receptor platform and a molecular binding system. Binders recognising cancer cells are administered intravenously after the administration of CAR-T cells and activates these cells resulting in on-demand tumour killing.
PTX is nearing completion of pre-clinical development of CellPryme and is preparing regulatory packages for both CellPryme-M and CellPryme-A so that these may enter the clinic in due course. The advantage of these platforms is that they can integrate easily into partner programs without highly disruptive changes to manufacturing processes or protocols mid-stream.
PTX is progressing platform optimisation of OmniCAR to investigate unarmed T-cell activity and improving control features. As a unique and multi-modal platform, this program is involving domain experts across protein and cell engineering and other areas.
This company is grossly undervalued
Currently Prescient stock is seriously undervaluing a potential near-term payday with PTX-100. Prescient Therapeutics had a market capitalisation on ASX of less than A$30m. we think the current market capitalisation of Prescient does not begin to take account of the way in which clinical success with PTX-100 in T-Cell Lymphoma can yield a marketed drug in only around three years, where that drug’s market opportunity is at least in the hundreds of millions.
Our friends at Pitt Street Research have been covering the company since November 2023, and most recently published a note on it earlier this week. Pitt Street valued the company at 11c per share in a base case and 16c per share in an optimistic (or bull) case. We encourage readers interested in hearing more about the company to check out Pitt Street’s reports for free.
Prescient is an exciting company that investors should keep an eye on. There are very few (if any) biotechs fighting cancer with such optimistic data and about to enter Phase 2.
Prescient Therapeutics is a research client of Pitt Street Research.
Blog Categories
Get the Latest Insider Trades on ASX!
Recent Posts
Duratec (ASX: DUR) Acquires Pacific Welding Australia for A$12 Million: Is the Stock a Buy Now?
Duratec Expands With A$12 Million Pacific Welding Deal Duratec (ASX: DUR) climbed 1.48% on Thursday after announcing it will buy…
China’s Manufacturing PMI Just Hit Its Best Level in a Year: These 5 ASX Stocks Look Like Buys Right Now
ASX Stocks to Buy: 5 Picks to Benefit from China’s PMI Surge China’s official NBS Manufacturing PMI climbed to 50.4…
KMD Brands (ASX: KMD) Crashes Over 50% After NZ$65 Million Raise at Nearly 70% Discount: Buy the Dip or Stay Away?
KMD Brands’ 70% Discounted Capital Raise: What It Means for Investors KMD Brands (ASX: KMD) lost more than half its…


