PolyNovo (ASX:PNV) gets another FDA green light

Polynovo (ASX: PNV) shareholders woke up to news that the FDA has cleared NovoSorb MTX for soft tissue regeneration for the management of complex wounds. But didn’t PolyNovo already have FDA approval? And what will this latest approval mean for its share price?
No time to do stock research, but you still want to invest?
Stocks Down Under Concierge gives you timely BUY and SELL alerts on ASX-listed stocks!
GET A 3-MONTH FREE TRIAL TO CONCIERGE TODAY
A good product, but COVID brings its run to a halt
Polynovo first won FDA approval for NovoSorb in 2015. It is a 2mm thick biodegradable polymer foam wound scaffolding that helps ailments where the body may struggle to rebuild its own cells, particularly severe burns and wounds.
As big a step as that FDA approval was, it was just the beginning given that burns were just 10% of its target market. PolyNovo wanted to expand into new target markets, including hand surgeries, hernia repair and breast implants, but had difficult times during the pandemic. Hospitals in the northern hemisphere were full of COVID-19 patients, leaving little room for sales, surgeries or clinical trials.
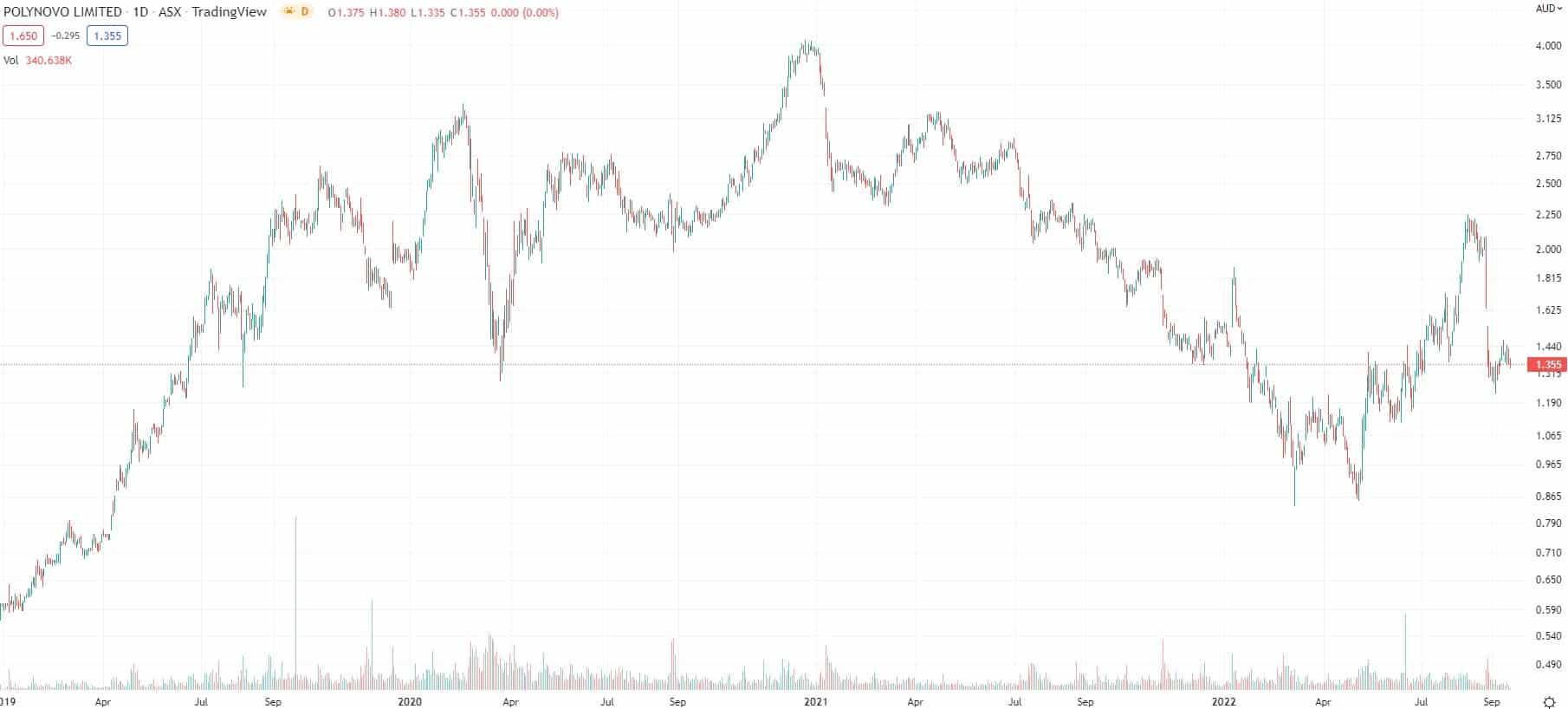
Polynovo (ASX:PNV) share price chart (Graph: TradingView)
A new dawn for PolyNovo?
This morning, PolyNovo announced it received FDA 510(k) clearance for NovoSorb MTX. This is an enhancement of the first version of NovoSorb, allowing NovoSorb to be used in procedures where the sealing membrane was not required.
A sealing membrane is one of the three layers of NovoSorb that acts as an interface for deep burns down to the muscle. The company estimates that this expanded approval increases its addressable market by A$500m.
What now?
In our view, this is not as big news as if the FDA had approved NovoSorb for additional indications with larger markets than chronic wounds. Nor is this bigger than if the FDA had begun by approving NovoSorb first for procedures where the sealing membrane was required followed by those where it was optional.
However, the FDA decision is nonetheless good for the company. It means that surgeons won’t have to ask whether or not a sealing membrane is required before even thinking about using NovoSorb. So, this announcement should do wonders for the share price!
No time to do stock research, but you still want to invest?
Stocks Down Under Concierge gives you timely BUY and SELL alerts on ASX-listed stocks!
GET A 3-MONTH FREE TRIAL TO CONCIERGE TODAY
No credit card needed and the trial expires automatically.

Blog Categories
Get the Latest Insider Trades on ASX!
Recent Posts
St George Mining (ASX:SGQ) 75% Resource Upgrade, Stock Down 15%, Why?
St George Mining Hits 70.9Mt, The Market Still Wants Confidence St George Mining announced a major resource upgrade at its…
Oil Supply Risk Rises After Iran Drone Attacks Reach Ras Tanura and UK Base in Cyprus
From Battlefield to Oil Chain, Drone Attacks Expand the Conflict Map A British military base at Akrotiri in Cyprus and…


